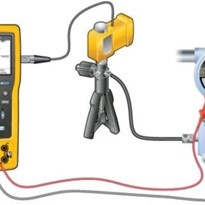
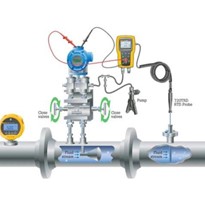
This article focuses on considerations for pressure calibrations using a high pressure hydraulic pump to generate the pressure.
Getting started
To produce stable and high pressure measurements using a hydraulic calibration pump, the gas within the calibration system needs to be removed.
Hydraulic test pumps use various types of fluids to generate high pressures. Because gas is much more compressible than liquid, purging most – if not all the gas – out of the system will allow for maximum pressures to be generated.
The following steps describe the procedure to purge the gas from an Additel test pump:
- Ensure the pump, reference standard, and device under test (DUT) are securely connected to the calibration pump.
- Close the vent valve and screw out the main screw press. You should see a vacuum being pulled on your reference and DUT(assuming the reference and the DUT are able to be used for vacuum measurement).
- Open the vent valve, wait for the pressure to settle to zero, and screw in the main screw press. As you do this, you may see bubbles emerge in the medium reservoir which is a good indication that gas is being pushed out of the system.
- Close the vent valve and repeat steps 2 and 3 one or two more times.
- Close the vent valve and unscrew the main screw press half way out. Then open the vent valve to zero the measurement.
Now, you are ready to close the vent valve and generate pressure.

Stable measurements
As pressures are generated to the desired test point it is common to initially observe a fairly rapid decrease in pressure.
Initially, you may conclude that this is a pressure leak but what you are likely observing is called the adiabatic effect. This effect is defined as a gain or loss of heat within a system and its environment.
When a gas is compressed under adiabatic conditions, its pressure increases and its temperature rises without the gain or loss of any heat. This happens when the screw press of a pump compresses the fluid volume, thus resulting in an increase in pressure but also an increase in the temperature. As the increase in pressure stops the temperature generated from the screw press dissipates.
If the volume is held constant and the temperature decreases so also will the pressure decrease. So this initial degrease of pressure is in fact a result of the temperature settling from the adiabatic heating effect generated from the screw press of the pump.

























-205x205.jpg)






